A New Robust Strength Enhancer Can Be Formulated With Existing Technologies to Improve Many Performance Aspects of Cement.
By Denise A. Silva and Richard Sibbick
While the ultimate strength of concrete (or compressive strength at 28 days) is the main parameter for the design of concrete structures, it is the strength that the concrete achieves up to 24 hours after placement that will typically define the speed of the construction. Therefore, early age strength enhancement is a much sought-after performance parameter by both the cement and the concrete producers.
Early age strengths are also critical to determine the maximum level of clinker replacement by a supplementary cementitious material (e.g. fly ash and slag) that the cement producer can afford to add. There are several parameters related to the physical and chemical/mineralogical properties of the cement that can be modified to increase its early age strength. Once these parameters are defined, chemical admixtures have been shown to play an important role to optimize the performance of the cement.
Strength enhancers have been extensively used by cement and concrete producers to boost the quality of their products. Some classes of strength enhancers can also prevent agglomeration of the fine cement particles during grinding, working as grinding aids. Examples of such chemicals are the alkanolamines, such as triethanolamine, diethanol isopropanolamine and tri-isopropanolamine, which have been developed by Cambridge, Mass.-based GCP Applied Technologies (formerly W.R. Grace) and extensively used by the cement industry for decades.
This article describes the performance and mechanism of a newly developed early strength enhancement technology, HES. The enhancer is a key ingredient in OPTEVA HE, a patent-pending family of strength enhancers from GCP Applied Technologies.
Strength Performance of New Grinding Aid
 |
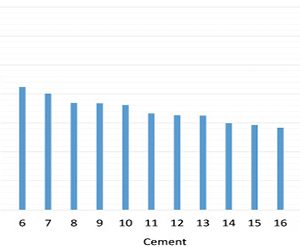
| Figure 1 – 1-day strength performance of the new strength enhancer HES in 23 cements. |
Figure 1 shows the strength performance at 24 hours of hydration of HES when added to the mixing water of EN-196 mortars prepared with ordinary portland cements (OPCs) from 23 different cement plants located in North America, Latin America and Europe. These are either commercial cements (produced in industrial mills) or cements ground in laboratory mills using commercial clinkers.
The strength results are expressed as a percent of the strength of each respective mortar in the absence of the additive. The active content of HES in the mortars ranged between 0.005 percent and 0.02 percent of the cement weight, and the results shown are the best response of the cement within this dose range. In other words, some cements showed a better response with lower dose while others reacted better with higher dose of the new chemical.
Considering an experimental error of 3 percent, 19 of the 23 OPCs tested (83. percent of the cements) showed up to 21 percent higher one-day strength than the reference mortar without additives, indicating a robust response of the technology across different cement chemistries.
In order to verify the impact of supplementary cementitious materials on the performance of the new additive, two OPCs were blended in a V-blender with 30 percent ground granulated blast furnace slag (GGBFS), 20 percent class F fly ash or 15 percent limestone (weight percent of OPC substitution), and EN-196 mortars were prepared to compare the early strengths in the presence and absence of 0.01 percent active HES (percent of cementitious weight).
The OPCs used in this experiment are Cement 3 from Figure 1 and a lab ground cement using the same clinker of Cement 1. These cements were chosen due to their good response to the new technology. Figure 2 shows the absolute compressive strength of the mortars at one day. It can be seen that the new technology is also able to substantially increase the early strength of blended cements.
Mechanisms of HES
 |
| (a) |
 |
| (b) |
| Figure 2 – 1-day strength performance in EN-196 mortars prepared with blends of (a) Cement 3 and (b) a lab ground cement using clinker from Cement 1, with the indicated supplementary cementitious materials. |
 |
| (a) |
 |
| (b) |
| Figure 3 – QXRD phase quantification of 24 hours old pastes prepared with (a) Cement 1 and (b) Cement 9. w/c ratio = 0.4. |
Mechanistic studies were conducted using two cements with different reactivity to the new technology, selected from Figure 1: Cement 1 is an industrial Type I cement according to the ASTM C150 classification (or CEM I according to the EN specification), and Cement 9 is a lab-ground cement using an ASTM C150 Type II/V clinker (corresponding to CEM I, but with low C3A content for higher sulfate resistance).
Pastes at water-to-cement (w/c) ratio of 0.4 were prepared with these two cements and distilled water for assessment of the impact of the new technology on cement hydration and microstructure after 24 hours of hydration. Quantitative x-ray diffraction (QXRD), mercury intrusion porosimetry (MIP) and optical microscopy of thin, polished sections were used for the investigation.
QXRD allows the identification and quantification of crystalline phases in polycrystalline materials, such as cementitious materials. The absolute content of anhydrous cement phases (alite, belite, C3A, C4AF) and of hydrated phases (calcium hydroxide and ettringite) can thus be determined to provide an insight on the impact of chemical additives on the cement hydration process.
MIP is one of the most common analytical techniques used for the characterization of the pore entry size distribution of porous materials including cement-based materials. Pore entries as small as 4 nm in diameter can be measured using this technique (Berodier et al, 2016). MIP is a helpful tool to investigate if a chemical additive induces changes in the porosity of the cement paste. Optical microscopic observation of thin sections using polarized light allows the observation of the distribution, amounts and locations of some relevant cement phases, such as residual cement grains (alite, belite and ferrite), hydration or reaction rims, calcium silicate hydrate (C-S-H), ettringite and calcium hydroxide, as well as an indication of the cement paste porosity distribution.
Figure 3 shows the QXRD phase quantification of the pastes prepared with Cements 1 and 9 at 24 hours of hydration. The error bars represent the standard deviations of pastes prepared from these cements on two different days.
The results show the only meaningful differences between the blank pastes and pastes with 0.02 percent active HES (percent of cement weight) are the contents of calcium hydroxide (portlandite) and ettringite: the new additive decreases the amount of portlandite and increases the amount of ettringite in cement pastes at 24 hours of hydration, without affecting the amount of alite and interstitial phases (C3A and C4AF). From these results, it appears that the new technology is changing the distribution of calcium (and perhaps aluminate and sulfate ions) in the paste.
For MIP, 2-mm thick disks were wet cut (using distilled water as the cooling agent) from the 0.4 w/c ratio paste specimens prepared with Cement 1 at 24 hours of hydration and immersed in isopropanol for a minimum of three days to remove the free water. The samples were then dried and kept under vacuum until testing. The distribution of the pore entry size of the samples is shown in Figure 4, where an overall reduction or refinement of the pore size distribution for diameters between 20 nm and 1 mm is observed with the new technology. This type of pore size refinement is a possible mechanism that could lead to the higher early strength observed when HES is used.
For the microscopy examination, the hydration of the pastes prepared with Cement 9 was interrupted at 24 hours by immersion in isopropanol, followed by vacuum impregnation of the sample with an ultra-low viscosity epoxy resin. Figure 5 shows optical microscopy images of ~25 micron thick thin sections of the pastes. Deposits of calcium hydroxide are clearly identified by the white color and needle-like shape crystals concentrated around hydrating cement grains. As can be seen in Figure 5b, the presence of 0.02 percent active HES (percent of cement weight) reduces the total amount of calcium hydroxide in the paste and promotes a better distribution and finer average crystal size of this phase in the paste. These results support the decrease in portlandite observed from the QXRD analysis reported in Figure 3.
The above results confirm a possible mechanism for strength enhancement of HES, which involves the refinement of the pore structure of the cement matrix. The impact on the amount and distribution of calcium hydroxide may also be a contributor to strength, as the impact on the reduction in size of the calcium hydroxide deposits may extend to the interfacial transition zone with aggregates. Further investigation on this aspect is on-going.
Grinding Performance
In addition to being a strength enhancer, HES also works as a grinding aid. Figure 6 shows the grinding performance of the new technology (expressed as Blaine specific surface area of the cement) as a function of the active dosage, in 150 minutes-long grinds performed in a laboratory batch ball mill at room temperature. The 5-kg ball mill capacity was loaded with 3.5 kg of clinker (96 percent weight) and natural gypsum (4 percent weight). As can be seen in this set of experiments, the Blaine surface area plateaus at 0.01 percent active of HES (percent of cement weight).
 |
| Figure 4 – MIP curves of 24 hours-old pastes prepared with Cement 1 (w/c = 0.4). Active content of HES is 0.02 percent on cement weight. |
 |
| (a) Blank |
 |
| (b) HES |
| Figure 5 – Optical microscopy of cement pastes (w/c = 0.4) prepared with Cement 9, in polished thin sections under crossed polarized light. Hydration age is 24 hours. Image widths 330 microns. |
 |
| Figure 6 – Blaine surface area of batch lab grinds of 150 minutes of duration performed at room temperature. |
 |
| (a) Blank |
 |
| (b) HES |
| Figure 7 – Aspect of mortars prepared according to Härdtl et al’s protocol for brown discoloration potential. |
Yellow Staining of Concrete
Brown/yellow discoloration of the concrete surface can occur when iron compounds migrate to and precipitate at the surface during the curing period. The chemistry of the cement, the nature of the aggregates, curing conditions, and high dosages of chemicals which chelate iron may elevate the concentration of iron in the pore water.
HES works as a grinding aid and strength enhancer without increasing the tendency of mortar and concrete to show yellow staining because it does not increase the solubility of iron. Figure 7 shows pictures of mortars prepared according to the protocol described by Härdtl et al (2003), by which mortars of high water/cement ratio and a graded sand are prepared and placed in polyethylene bags.
Slits are cut on the corners of the bags at 7 days of hydration, and visual observations and chromatometry measurements made at 28 days of hydration allow the identification and quantification of the intensity of brown/yellow staining. As can be seen in the images taken at the slits region, the new technology does not cause any change in the color of the mortar as compared to the blank mortar.
Summary
HES is a new robust early strength enhancer that also works as a grinding aid and can be formulated with existing technologies to improve many performance aspects of the cement. Mechanistic investigations showed that the strength enhancement is a result of a refinement of the pore structure of the cement matrix.
In addition, HES does not induce any brown/yellow staining on the surface of finished mortar and concrete. It also does not have any impact on water reduction, air entrainment and the quality of the air void system in concrete made with different concrete admixtures, such as air entrainers and water reducers.
Denise A. Silva and Richard Sibbick are with GCP Applied Technologies (formerly W.R. Grace).